 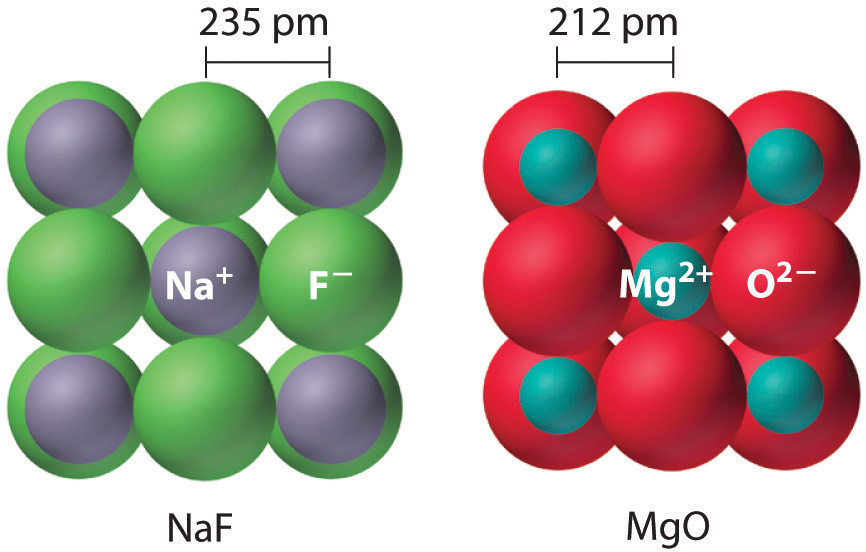 
Dutton, Ultraviolet absorption of alkali halides Phys.Rev. Gurney, "Electronic processes in Ionic crystals", 2nd. Fowler, "Physics of Color Centers", New York: Academic Press (1968) Journal de Physique et le Radium, 28 (1981).Elastic constants of the Alkali Halides at 4.2K Phys.Rev. New high pressure polymorphs Sodium halides, T.Spin-orbit coupling: 0.52 ( valence band) (Ref.5) Polaron coupling constant: a = 5.0 ( for m*=1 )Įlectron affinity: ( in eV., from bottom of conduction band under vacuum)ģ.51, by Mott-Gurney, maybe 3eV. Lipari, Electronic structure of NaBr, Phys.Rev. II: Critical survey of theoretical calculation, R.T. Transverse optic phonon T0 (k=0): 134 or 146 cm -1Įlectronic band structure of the alkali halides. Where all the numbers you plug in are positive.Stiffness constants: in 10 11 dynes/cm 2, at room temperatureĬompressibility (in 10 11 dynes/cm 2): 0.5023 ? 
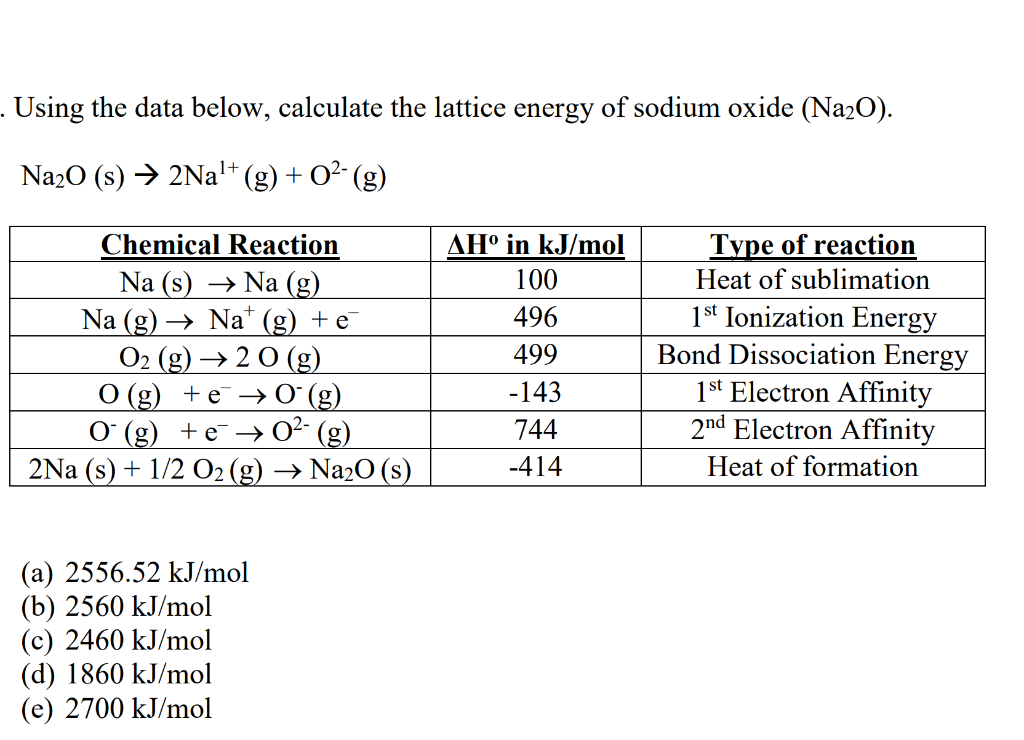
#color(blue)(DeltaH_"lattice" -= -|DeltaH_"lattice"|)# Solving for #DeltaH_"lattice"# usually gives a positive answer, so we take the negative of the answer by convention to get: The polymer electrolyte system of methylcellulose (MC) doped with various sodium bromide (NaBr) salt concentrations is prepared in this study using the solution. Take the step as being upwards to generate a complete cycle, for which #DeltaH_"cycle" = 0# (since #H_f = H_i# for a complete cycle). #"These cancel out completely upon adding, proving"#Īnd now if we wish, the lattice energy can be calculated. Put this all together, with some data, and we get, for #"1 mol"# of #"NaCl"(s)#: 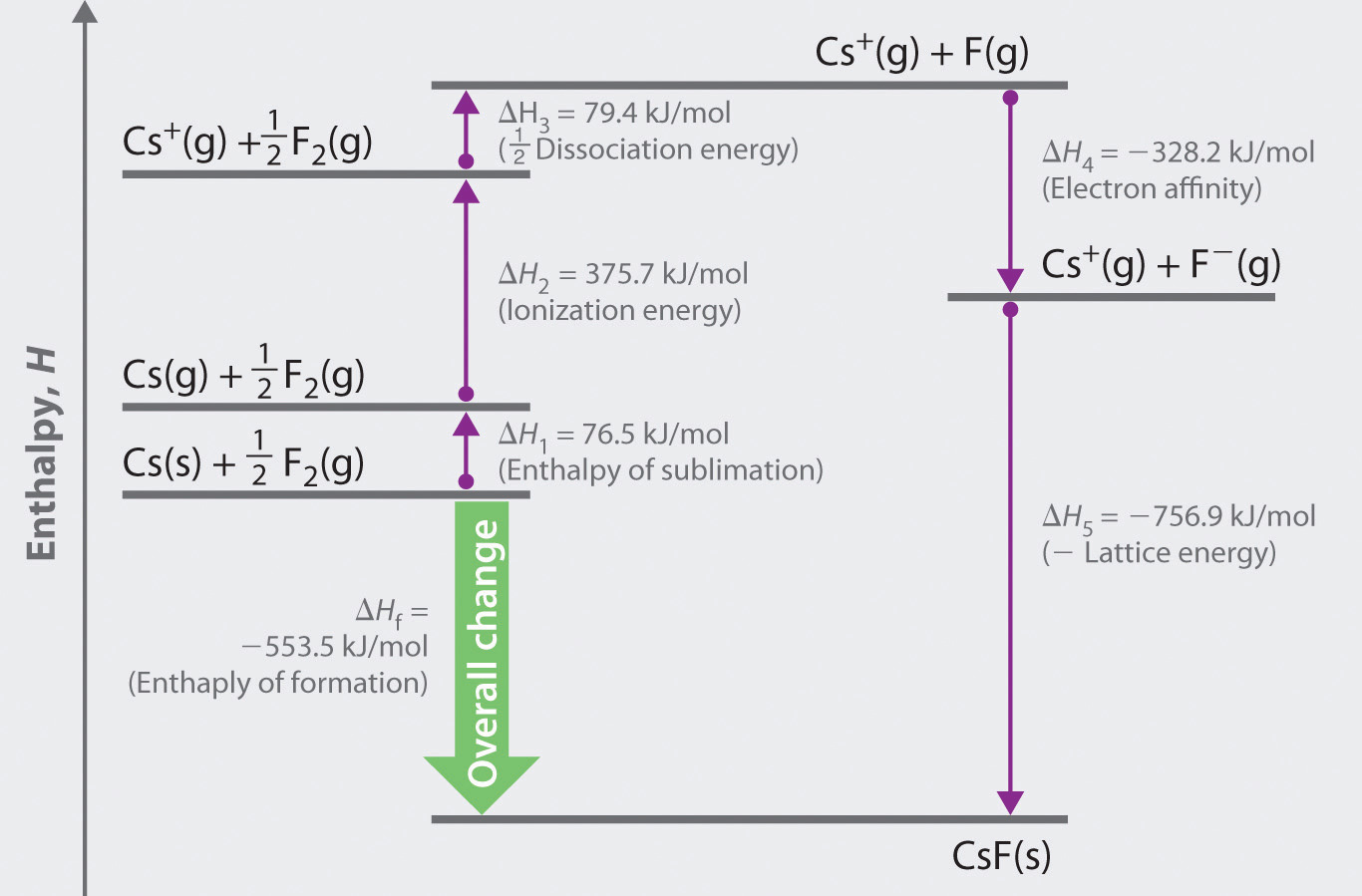
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
